
The stronger a bond, the greater the energy required to break it. Separating any pair of bonded atoms requires energy (see Figure 1 in Chapter 7.2 Covalent Bonding). We measure the strength of a covalent bond by the energy required to break it, that is, the energy necessary to separate the bonded atoms. Stable molecules exist because covalent bonds hold the atoms together. In the other type of bond known as a covalent bond, there is a sharing.
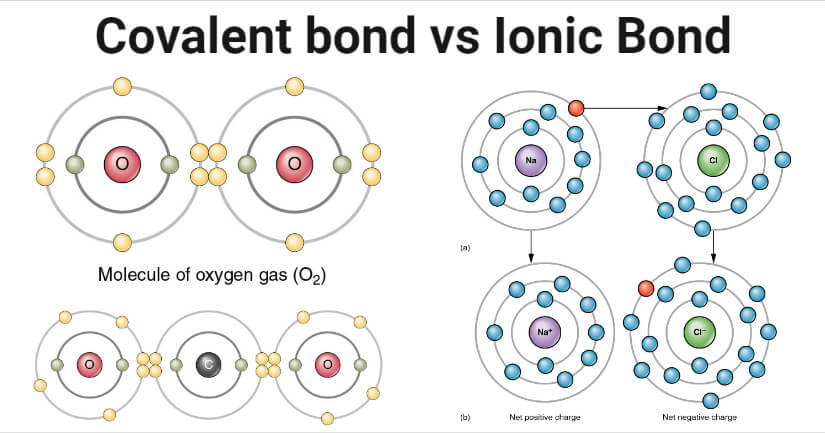
In this section, you will learn about the bond strength of covalent bonds, and then compare that to the strength of ionic bonds, which is related to the lattice energy of a compound. More the charge disparity between the cation and the anion, the stronger the ionic bond.

By the end of this section, you will be able to: So what answer should you give for this class Because this is a biology class, you should say that covalent bonds are stronger than ionic bonds because they.
